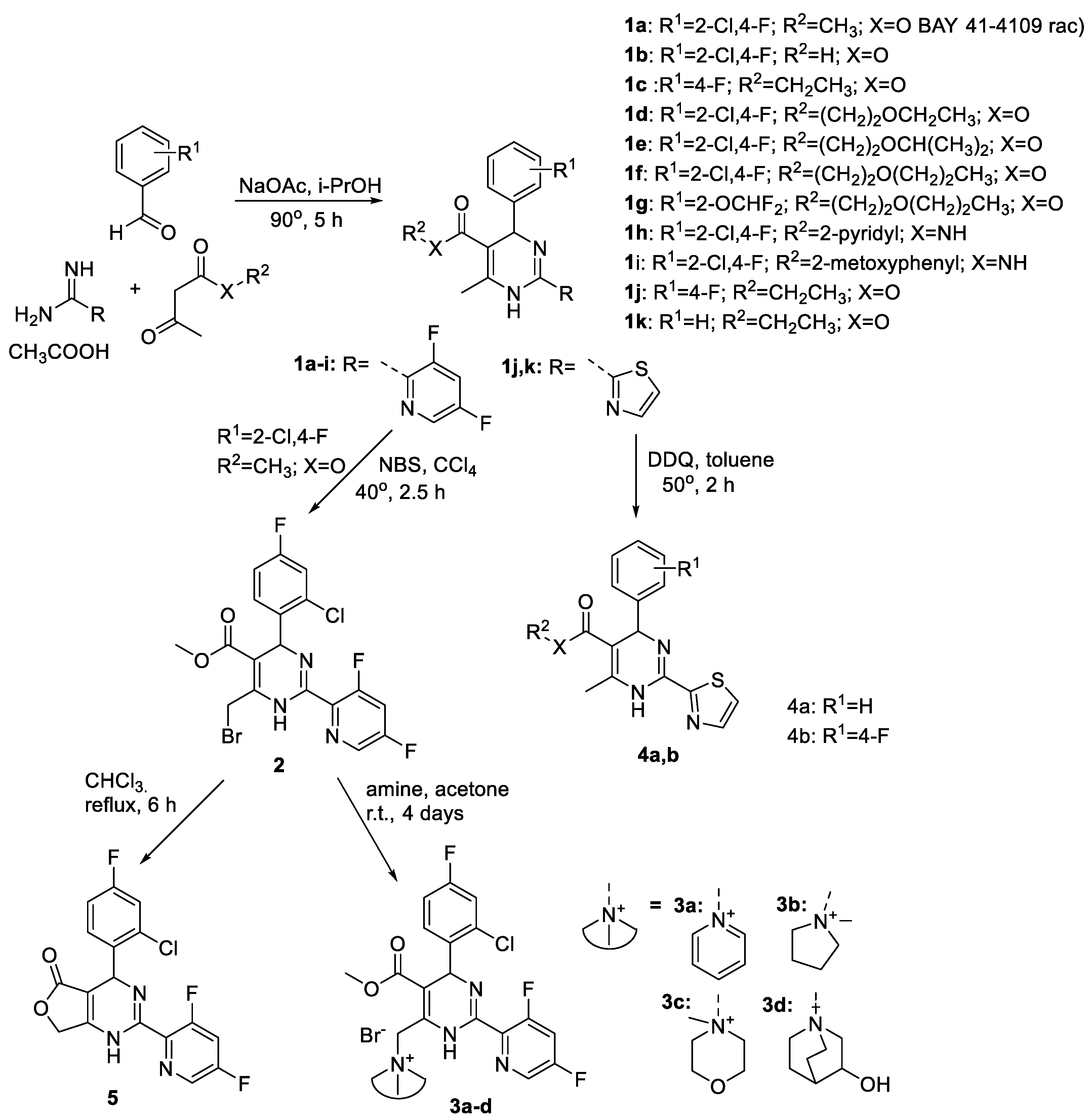
Structural Modification of Natural Product Ganomycin I Leading to Discovery of a α-Glucosidase and HMG-CoA Reductase Dual Inhibitor Improving Obesity and Metabolic Dysfunction in Vivo | Journal of Medicinal Chemistry
![TfOH-Catalyzed Cascade C–H/N–H Chemo-/Regioselective Annulation of Indole-2-carboxamides with Benzoquinones for the Construction of Anticancer Tetracyclic Indolo[2,3-c]quinolinones | The Journal of Organic Chemistry TfOH-Catalyzed Cascade C–H/N–H Chemo-/Regioselective Annulation of Indole-2-carboxamides with Benzoquinones for the Construction of Anticancer Tetracyclic Indolo[2,3-c]quinolinones | The Journal of Organic Chemistry](https://pubs.acs.org/cms/10.1021/acs.joc.2c00598/asset/images/medium/jo2c00598_0004.gif)
TfOH-Catalyzed Cascade C–H/N–H Chemo-/Regioselective Annulation of Indole-2-carboxamides with Benzoquinones for the Construction of Anticancer Tetracyclic Indolo[2,3-c]quinolinones | The Journal of Organic Chemistry

Selenylation Chemistry Suitable for On‐Plate Parallel and On‐DNA Library Synthesis Enabling High‐Throughput Medicinal Chemistry - Xu - Angewandte Chemie International Edition - Wiley Online Library
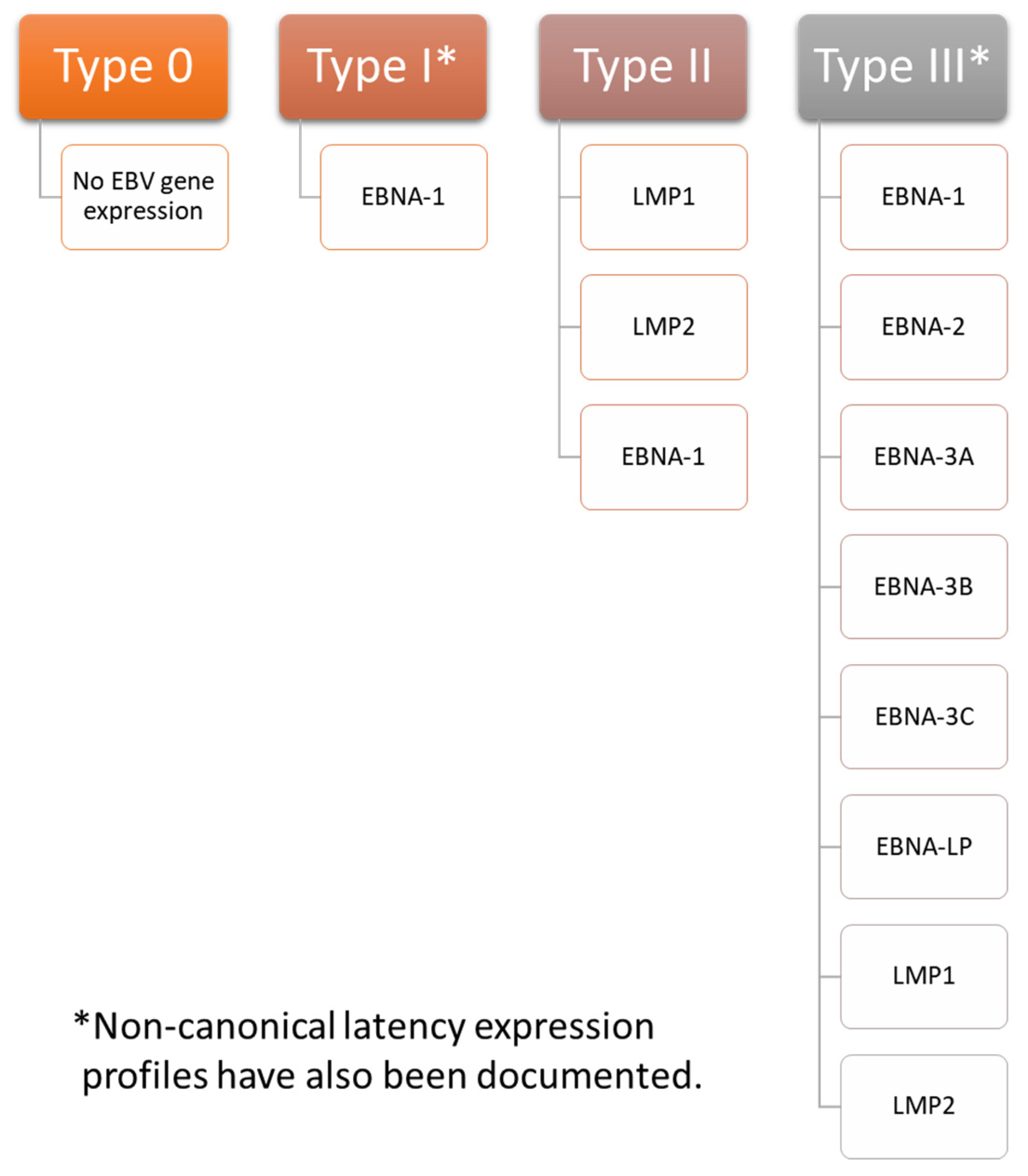
Pharmaceuticals | Free Full-Text | Design and Synthesis of Hepatitis B Virus (HBV) Capsid Assembly Modulators and Evaluation of Their Activity in Mammalian Cell Model | HTML

The Alpha Keto Amide Moiety as a Privileged Motif in Medicinal Chemistry: Current Insights and Emerging Opportunities | Journal of Medicinal Chemistry
A Randomized Phase I Study to Evaluate the Safety, Tolerability, and Pharmacokinetics of Recombinant <i>Erwinia</i>
Pharmaceuticals | Free Full-Text | Antimicrobial Activity of Rhenium Di- and Tricarbonyl Diimine Complexes: Insights on Membrane-Bound S. aureus Protein Binding | HTML

On-Demand Activation of a Bioorthogonal Prodrug of SN-38 with Fast Reaction Kinetics and High Releasing Efficiency In Vivo | Journal of Medicinal Chemistry

Isatin Compounds as Noncovalent SARS Coronavirus 3C-like Protease Inhibitors | Journal of Medicinal Chemistry